 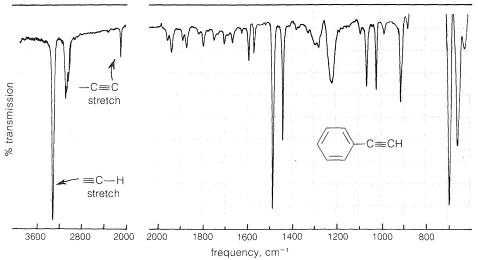
Stretching absorptions are marked in blue, bending absprptions in green. The stretching vibration of the CC bond usually gives rise to a moderate band in the region. The bands at 917 cm -cm -1 are attributed to =C&Mac173 H bends. The use of infrared spectroscopy for determining the substitution pattern of alkenes is illustrated by the following data, and the spectra examples underneath. Alkenes are compounds that have a carbon-carbon double bond, CC. Bands for CH scissoring (1465) and methyl rock (1378) are marked on this spectrum in routine IR analysis, these bands are not specific to an alkene and are generally not noted because they are present in almost all organic molecules (and they are in the fingerprint region). Generally, asymmetric vibrational stretch frequency of alkene C-H is around 3150 cm-1. This study depicts the use of a fiber-optic coupled Fourier transform infrared spectroscopy-attenuated total reflection (FTIR-ATR) probe for the in-depth. In the region of 4000 - 2000 cm 1, the appearance of absorption bands usually comes from stretching vibrations between hydrogen and other atoms. Note the band greater than 3000 cm -1 for the =CH stretch and the several bands lower than 3000 cm -1 for ≬H stretch (alkanes). For systematic evaluation, the IR spectrum is commonly divided into some sub-regions. The IR spectrum of 1-octene is shown below. These bands are in the region 1000-650 cm -1 (Note: this overlaps the fingerprint region). 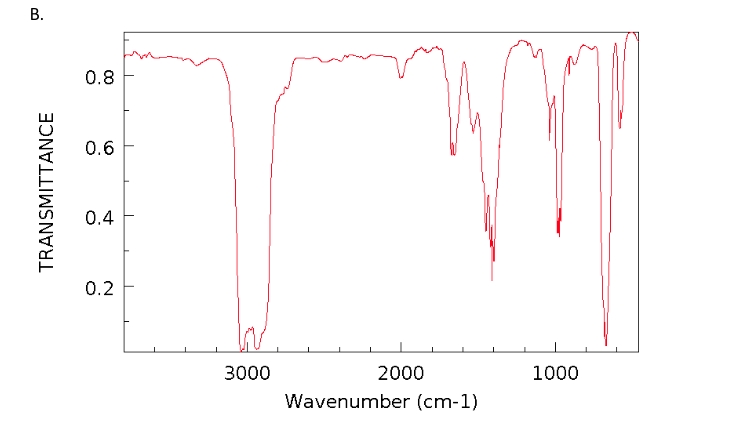
As you read the spectrum of benzene from left to right, note there are three peaks between 32 cm-1, making these C-H stretches. In 1 H NMR spectroscopy, the hydrogen bonded to the carbon adjacent to double bonds will give a H of 4.56.5 ppm. Figure 3: The IR spectrum of benzene, measured as a capillary thin film between two KBr windows. The stretching of CC bond will give an IR absorption peak at 16701600 cm 1, while the bending of CC bond absorbs between 1000 and 650 cm 1 wavelength. The strongest bands in the spectra of alkenes are those attributed to the carbon-hydrogen bending vibrations of the = CH group. The use of infrared spectroscopy for determining the substitution pattern of alkenes is illustrated by the following data, and the spectra examples underneath. The IR spectrum of benzene is shown in Figure 3. Compounds that do not have a C=C bond show C-H stretches only below 3000 cm -1. This is a very useful tool for interpreting IR spectra: Only alkenes and aromatics show a C-H stretch slightly higher than 3000 cm -1. The IR Spectrum Table is a chart for use during infrared spectroscopy. 
Stretching vibrations of the ≬= CH bond are of higher frequency (higher wavenumber) than those of the ≬ CH bond in alkanes. The stretching vibration of the C=C bond usually gives rise to a moderate band in the region 1680-1640 cm -1. IR: alkenes IR Spectroscopy Tutorial: AlkenesĪlkenes are compounds that have a carbon-carbon double bond, ≬=C. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
